Aufbau Principle & Filling Order
Questions 1–4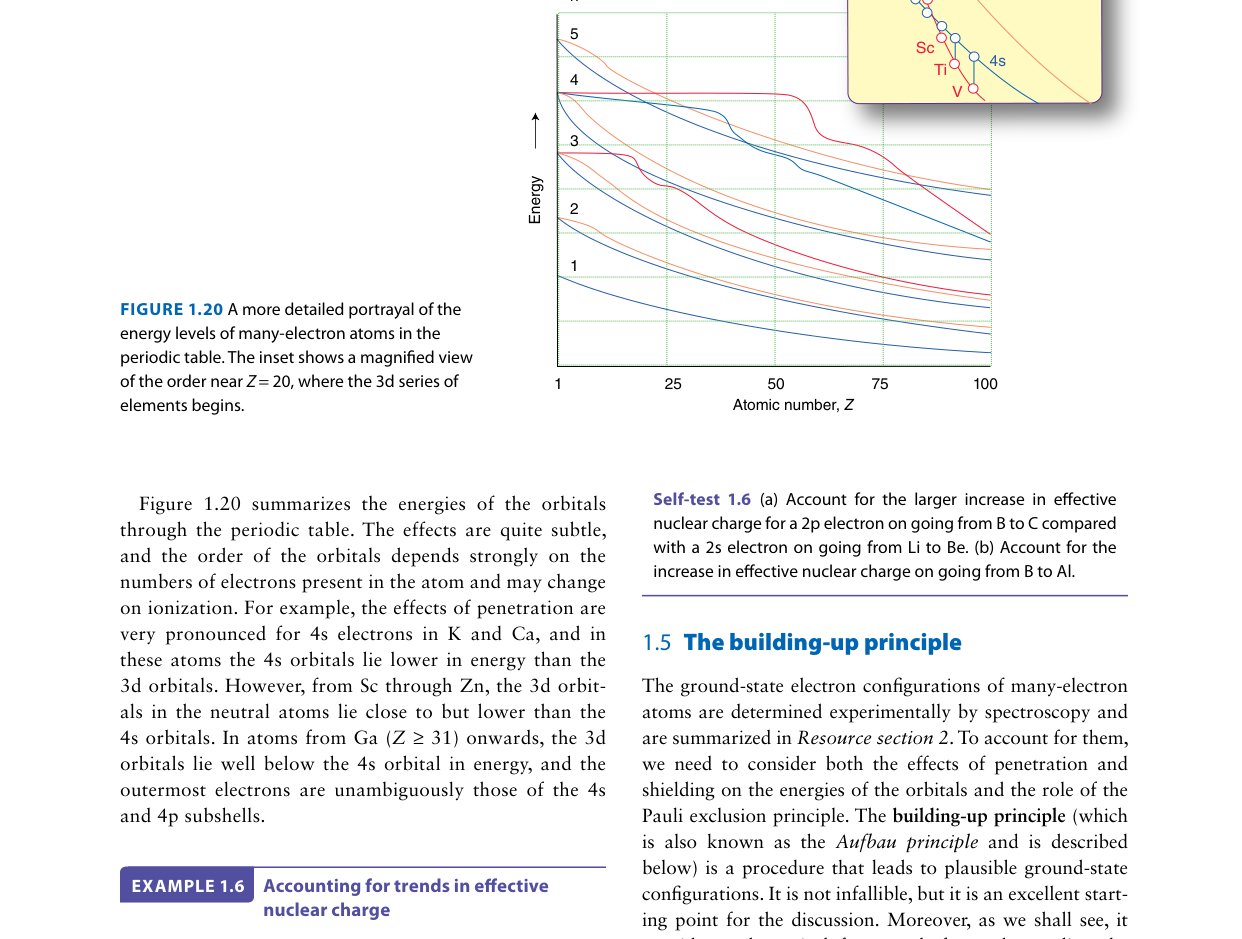
According to the Madelung (n+l) rule shown in the filling diagram above, which subshell fills first: 4s or 3d?
The Madelung rule states that subshells fill in order of increasing (n+l). When two subshells have the same (n+l) value, the one with lower n fills first. For 4s: n+l = 4+0 = 4. For 3d: n+l = 3+2 = 5. Since 4 < 5, the 4s subshell fills before 3d. This is why potassium (Z=19) has the configuration [Ar] 4s1 rather than [Ar] 3d1. Note, however, that this rule describes filling order for neutral atoms — once electrons are present, the actual orbital energies can cross, which is why 4s electrons are lost before 3d when forming cations.

Chromium has the ground-state configuration [Ar] 3d5 4s1 rather than the expected [Ar] 3d4 4s2. Which factor best explains this anomaly?
The exchange energy is a quantum mechanical stabilization that arises when electrons with parallel spins occupy different orbitals of the same subshell. A half-filled d5 configuration (all five d orbitals singly occupied with parallel spins) maximizes the number of exchange pairs: K = n(n−1)/2 = 5(4)/2 = 10 pairs. The d4 configuration gives only 4(3)/2 = 6 pairs. This gain of 4 additional exchange pairs provides enough stabilization to offset the cost of promoting one electron from the 4s to the 3d subshell. The 4s and 3d energies are very close in chromium, making this promotion energetically favourable.
Copper has the ground-state configuration [Ar] 3d10 4s1 instead of the predicted [Ar] 3d9 4s2. Which explanation best accounts for this?
The fully-filled 3d10 configuration maximizes both exchange energy and the symmetrical distribution of electron density in the d subshell. With 10 electrons occupying all five 3d orbitals, there is no net orbital angular momentum contribution from the d shell, leading to a spherically symmetric charge distribution. Combined with the fact that by Z = 29 the 3d orbitals have dropped well below 4s in energy, the additional stabilization of the complete d shell easily compensates for the cost of having only one 4s electron. The 3d9 4s2 configuration is not forbidden but is simply higher in total energy.
Carbon has the configuration 1s2 2s2 2p2. According to Hund's rule, which arrangement of the two 2p electrons represents the ground state?
Hund's rule of maximum multiplicity states that for a given electron configuration, the term with the greatest spin multiplicity (i.e. the most unpaired electrons with parallel spins) has the lowest energy. For carbon's two 2p electrons, this means each electron occupies a separate 2p orbital with parallel spins: (↑)(↑)( ). This gives S = 1 and a spin multiplicity of 2S+1 = 3 (a triplet state, specifically 3P0). The parallel-spin arrangement minimizes electron–electron repulsion because electrons with the same spin are kept apart by the exchange interaction (Fermi hole), lowering the overall energy.
Electron Configurations of Ions
Questions 5–8
Iron has the neutral atom configuration [Ar] 3d6 4s2. When forming Fe2+, which electrons are removed first, and what is the resulting configuration?
Although the 4s orbital fills before 3d in neutral atoms (aufbau order), the 4s electrons are always removed first during ionization of d-block elements. This is because once the 3d subshell is occupied, the 3d orbitals contract significantly and drop below 4s in energy. The 4s electrons, being in a higher-energy orbital in the cation, are lost first. Consequently, Fe2+ = [Ar] 3d6, with six 3d electrons remaining. This principle applies universally: for any transition metal cation, remove ns electrons before (n−1)d electrons.
Titanium has the configuration [Ar] 3d2 4s2. What is the electron configuration of the Ti3+ ion?
Following the rule that ns electrons are removed before (n−1)d electrons in transition metal cations: Ti → Ti+ removes one 4s electron ([Ar] 3d2 4s1), Ti+ → Ti2+ removes the second 4s electron ([Ar] 3d2), and Ti2+ → Ti3+ removes one 3d electron ([Ar] 3d1). The Ti3+ ion is a d1 system with one unpaired electron, making it paramagnetic and giving it a characteristic purple colour in aqueous solution ([Ti(H2O)6]3+).
Compare Fe2+ ([Ar] 3d6) and Fe3+ ([Ar] 3d5). Which ion has more unpaired electrons?
Applying Hund's rule to the free (gas-phase) ions: Fe3+ has 5 d-electrons distributed as (↑)(↑)(↑)(↑)(↑) — one in each of the five 3d orbitals, all with parallel spin — giving 5 unpaired electrons. Fe2+ has 6 d-electrons: five orbitals singly occupied plus one doubly occupied, i.e. (↑↓)(↑)(↑)(↑)(↑), giving 4 unpaired electrons. Therefore Fe3+ (5 unpaired) has more than Fe2+ (4 unpaired). This is for the free ion; in coordination compounds, the crystal field can alter the number of unpaired electrons depending on whether the complex is high-spin or low-spin.
In the lanthanides, the 4f subshell fills before the 5d subshell for most elements. However, cerium (Ce, Z=58) has the configuration [Xe] 4f1 5d1 6s2 rather than [Xe] 4f2 6s2. Which statement best explains this?
At the beginning of the lanthanide series, the 4f and 5d orbitals are extremely close in energy. For cerium, the interelectronic repulsion cost of placing two electrons in the compact 4f subshell outweighs the small orbital energy difference. By distributing one electron each in 4f and 5d, the atom minimizes electron–electron repulsion while also benefiting from exchange interactions across both subshells. As we proceed along the series (Pr onward), the 4f orbitals drop further in energy due to the increasing nuclear charge, and the 5d is no longer occupied — giving configurations like [Xe] 4f3 6s2 for Pr. Lanthanum (Z=57) itself has [Xe] 5d1 6s2 with no 4f electrons at all, showing the crossover point.
Effective Nuclear Charge
Questions 9–11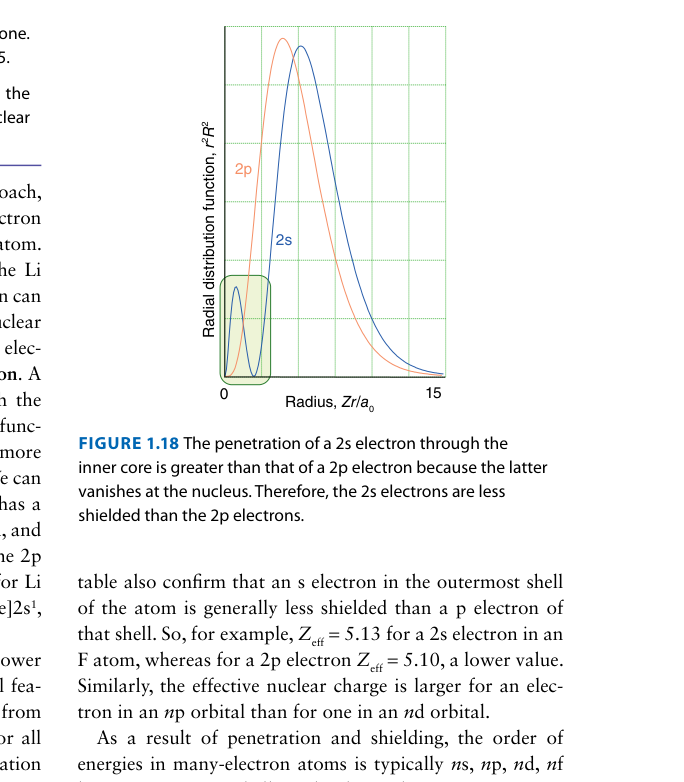
Use Slater's rules to estimate Zeff for a 2p electron in oxygen (Z = 8, configuration 1s2 2s2 2p4). Group the electrons as (1s2)(2s2 2p4). Each same-group electron shields by 0.35 and each 1s electron shields by 0.85. What is Zeff?
Using Slater's rules for a 2p electron in oxygen: we do not count the electron itself in the shielding sum. The (2s, 2p) group has 6 electrons total; excluding the one we are calculating Zeff for, there are 5 remaining same-group electrons, but wait — we must be precise. Slater groups (2s, 2p) together as one group. For a 2p electron: same-group contribution = 4 other electrons in (2s2 2p4) minus the electron itself = 5 × 0.35? Let us recount. Oxygen has 8 electrons: (1s2)(2s22p4). The electron of interest is one of the 2p electrons. Same-group (2s,2p) electrons excluding itself = 5. But the correct Slater calculation recognizes that the 2s22p4 group has 6 electrons; excluding the one of interest leaves 5 × 0.35 = 1.75. The (1s2) electrons each contribute 0.85: 2 × 0.85 = 1.70. So S = 1.75 + 1.70 = 3.45, giving Zeff = 8 − 3.45 = 4.55. However, option (b) uses 4 same-group electrons at 0.35 (a common textbook simplification where only the other 2p electrons are counted as same-group for the 2p electron, not the 2s). In many textbook treatments Slater groups (2s,2p) together giving answer (a). The answer (b) = 4.90 uses the alternative counting where only 4 electrons shield at 0.35 and 2 at 0.85. Both methods appear in different textbooks. The key concept is that Zeff is substantially less than Z = 8 due to shielding, with inner-shell electrons shielding far more effectively than same-shell electrons.
As we move from left to right across a period, Zeff experienced by the valence electrons generally increases. What is the fundamental reason for this trend?
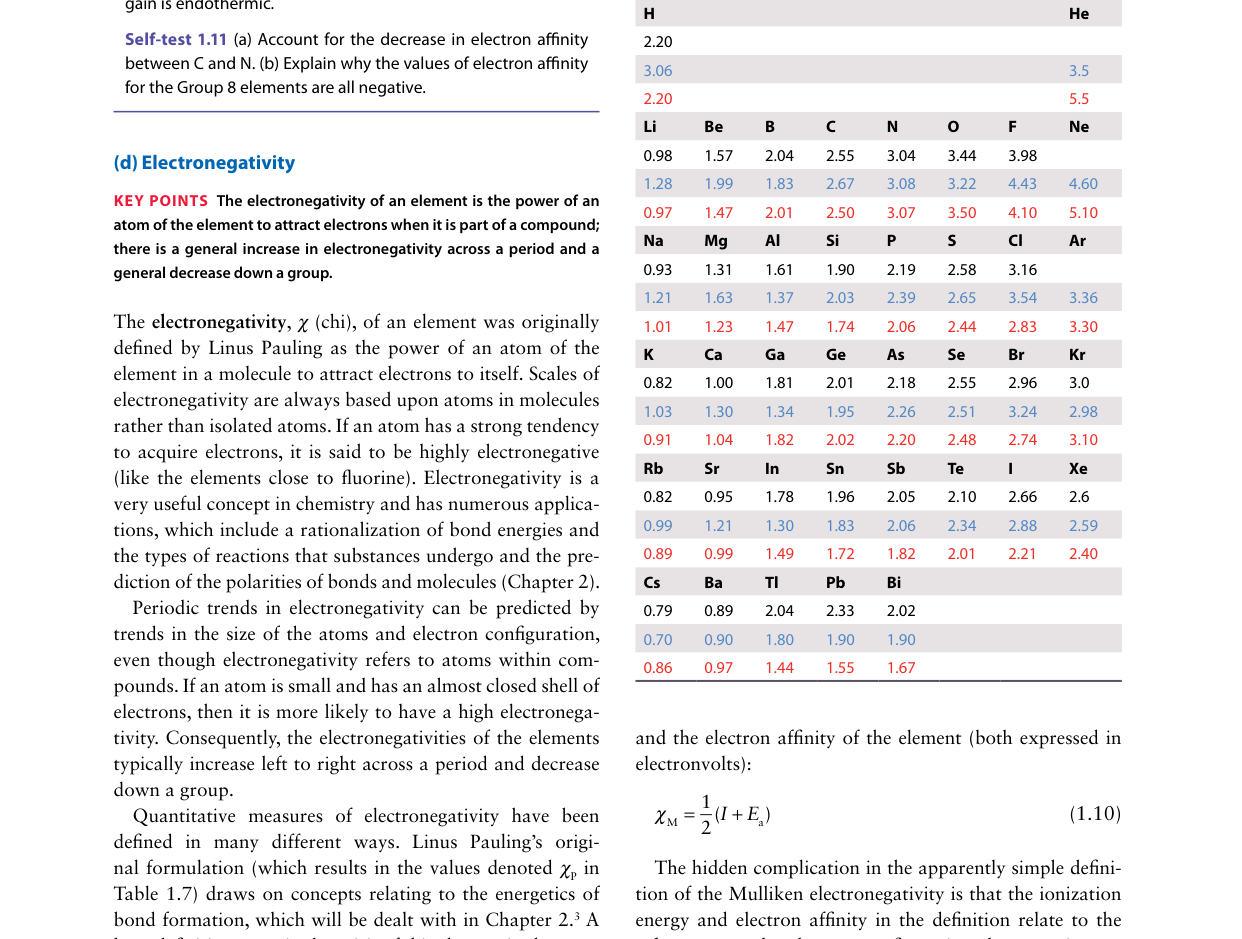
Across a period, each successive element has one additional proton in the nucleus and one additional electron in the same valence shell. According to Slater's rules, same-shell electrons shield each other by only about 0.35, while the new proton adds a full +1.0 to the nuclear charge. The net effect is an increase in Zeff of roughly 1.0 − 0.35 = +0.65 per element. This steadily increasing effective nuclear charge is the root cause of several periodic trends: decreasing atomic radius, increasing ionization energy, and increasing electronegativity across a period.
Place the following in order of increasing Zeff experienced by the outermost electron: Na (3s1), Mg (3s2), Al (3p1).
Using Slater's rules: Na (Z=11) 3s electron has Zeff ≈ 11 − 8.8 = 2.2. Mg (Z=12) 3s electron has Zeff ≈ 12 − 9.15 = 2.85. Al (Z=13) 3p electron has Zeff ≈ 13 − 9.50 = 3.50. The order is Na < Mg < Al. Even though Al's outer electron is in a 3p orbital (which penetrates less than 3s), the increase in nuclear charge from Z=11 to Z=13 more than compensates. This increasing Zeff across the period is the fundamental driver behind the decrease in atomic radius from Na to Al and the general increase in ionization energy.
Periodic Trends in Detail
Questions 12–15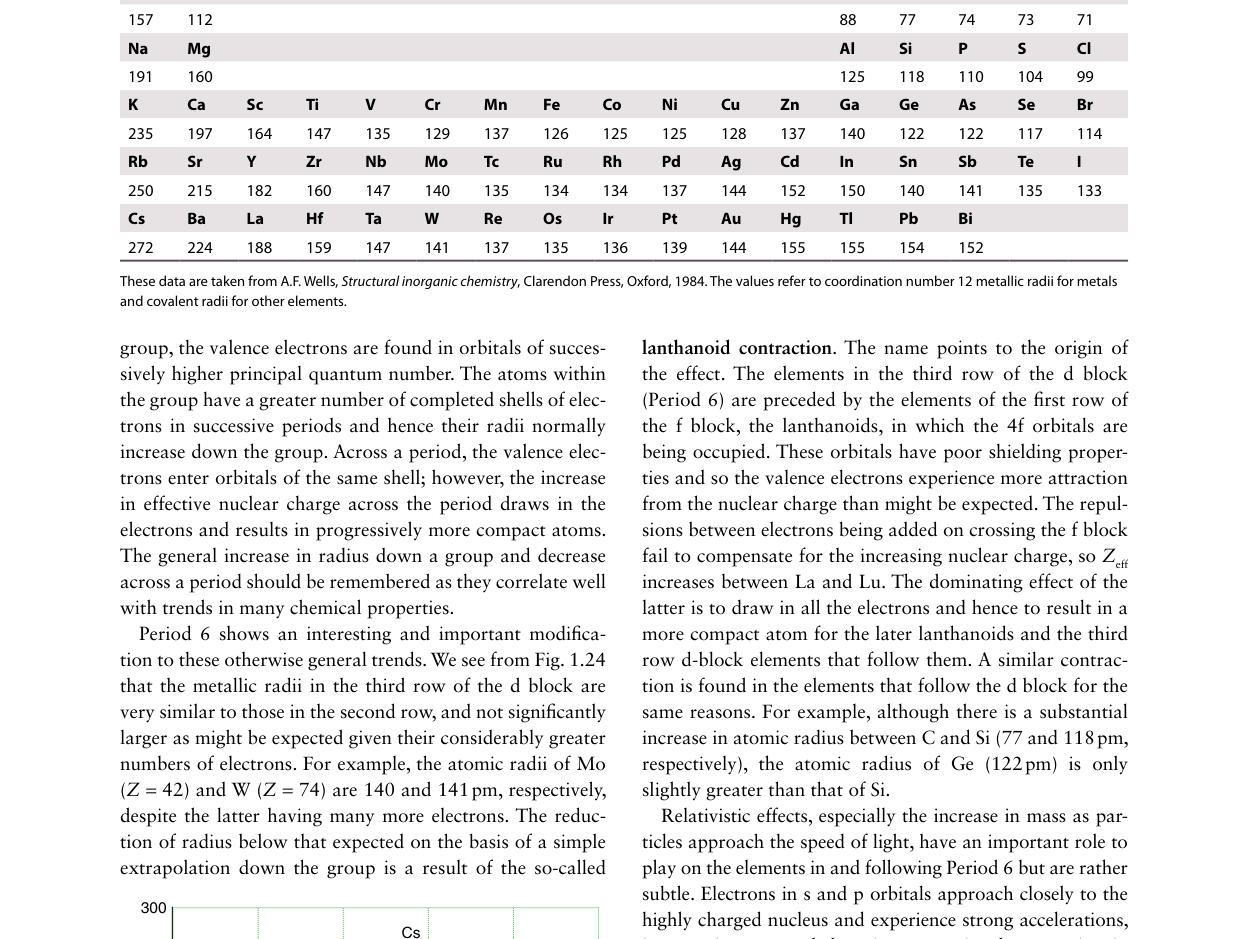
Examine the figure above. Across the d-block (e.g. Sc to Zn), atomic radii decrease only slightly compared with the sharp decrease across s- and p-blocks. What is the primary explanation?
In the d-block, each additional electron enters the (n−1)d subshell, which lies inside the ns valence shell. These d-electrons are relatively effective at shielding the outer ns electrons from the nuclear charge. In Slater's rules, (n−1) group electrons shield by ~0.85, compared with same-group electrons at only ~0.35. Consequently, as each proton is added (Z increases by 1), the d-electron added simultaneously provides significant shielding (~0.85), so Zeff felt by the ns electron increases only slightly (~+0.15 per element). This gentle increase in Zeff produces only a modest contraction in atomic radius across the d-block, in stark contrast to the s- and p-blocks where same-shell electrons shield poorly.
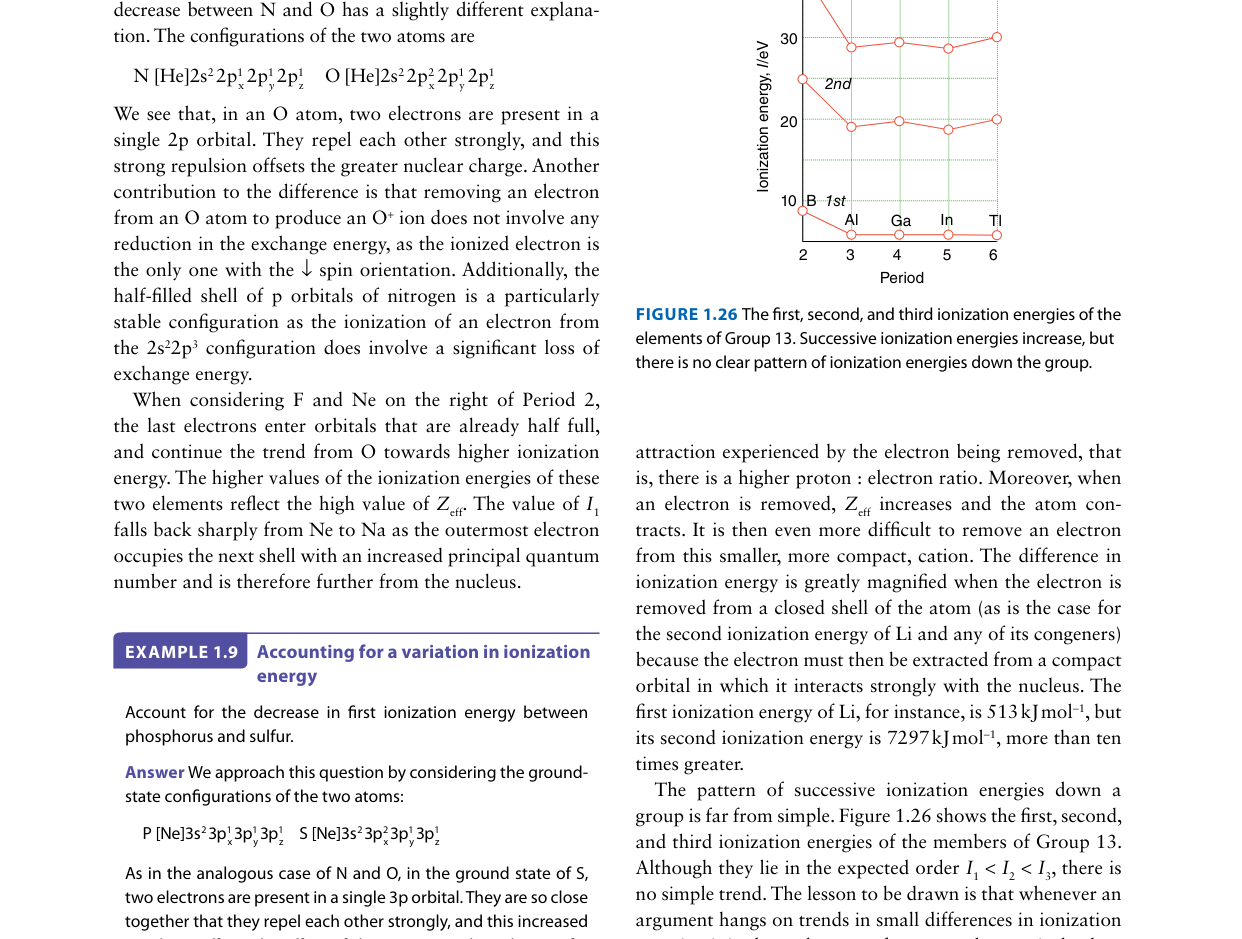
The first ionization energy of beryllium (Be, 900 kJ/mol) is higher than that of boron (B, 801 kJ/mol), even though boron has a greater nuclear charge. Which explanation accounts for this anomaly?
Beryllium (1s2 2s2) must lose a 2s electron upon ionization, while boron (1s2 2s2 2p1) loses its single 2p electron. The 2p orbital is higher in energy than 2s in a many-electron atom because 2s penetrates closer to the nucleus and experiences a higher Zeff. Therefore, the 2p electron in boron is more loosely bound and easier to remove, despite boron having Z = 5 versus Z = 4 for beryllium. Additionally, removing the 2p1 electron from boron leaves behind the stable, fully-filled 2s2 configuration, which provides no resistance. This is a classic example of how subshell structure causes deviations from the general trend of increasing IE across a period.
Fluorine is the most electronegative element, yet its electron affinity (EA = −328 kJ/mol) is less negative (less exothermic) than that of chlorine (EA = −349 kJ/mol). What is the best explanation?
Fluorine's very small atomic radius (the smallest of all halogens) means that its 2p orbitals are extremely compact. When an additional electron enters the 2p subshell to form F−, it must squeeze into a very small volume already occupied by 7 electrons (1s2 2s2 2p5). The resulting electron–electron repulsion is substantial and reduces the energy released upon electron attachment. Chlorine, being larger (3p orbitals are more spacious), has less interelectronic repulsion when gaining its extra electron, so more energy is released (EA is more negative). This is why chlorine, not fluorine, has the most negative electron affinity among all elements, despite fluorine having the higher electronegativity (electronegativity reflects the overall tendency to attract electrons in a bond, not just isolated electron attachment).
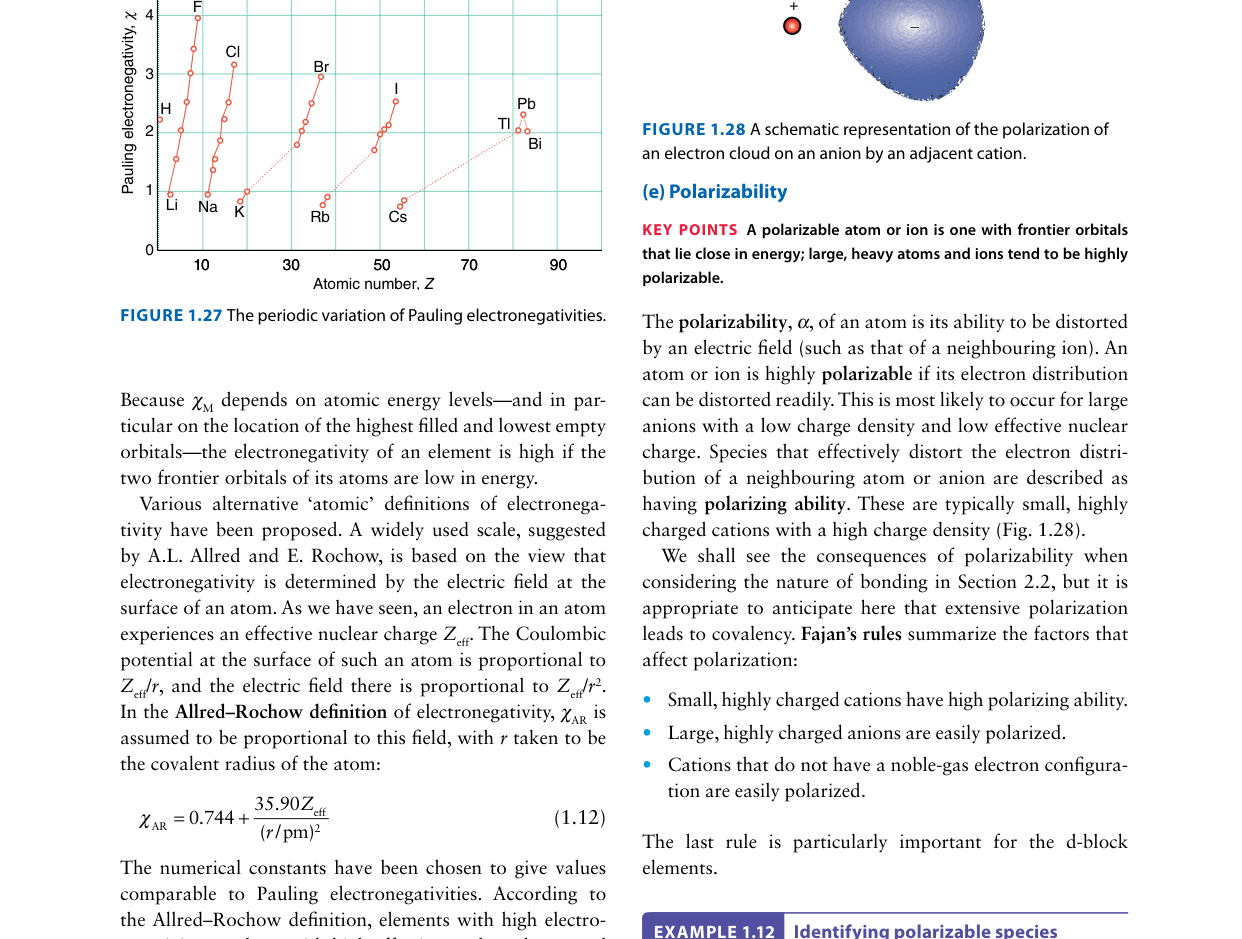
According to Fajans' rules, which combination of cation and anion properties leads to the greatest covalent character in an ionic compound?
Fajans' rules state that covalent character in an ionic compound increases when: (1) the cation is small and highly charged (high charge density → strong polarizing power), and (2) the anion is large and highly charged (large, diffuse electron cloud → high polarizability). A small cation like Li+ or Al3+ distorts the electron cloud of the anion, pulling it toward itself and creating a region of shared electron density — effectively introducing covalent character. For example, AlCl3 is significantly more covalent than NaCl because Al3+ is much smaller and more highly charged than Na+. Fajans' rules also predict that cations with non-noble-gas configurations (e.g. Cu+, with its 3d10 shell that shields the nucleus poorly) are more polarizing than similarly-sized noble-gas configuration cations (e.g. Na+).