Wavefunctions & Probability
Questions 1–3
According to the Born interpretation shown above, what physical meaning does ψ² (or |ψ|²) have at a given point in space?
The Born interpretation states that ψ² (or |ψ|² for complex wavefunctions) gives the probability density — the probability of finding the electron per unit volume at that point. Where ψ² = 0, there is zero probability of finding the electron; these locations are called nodes.
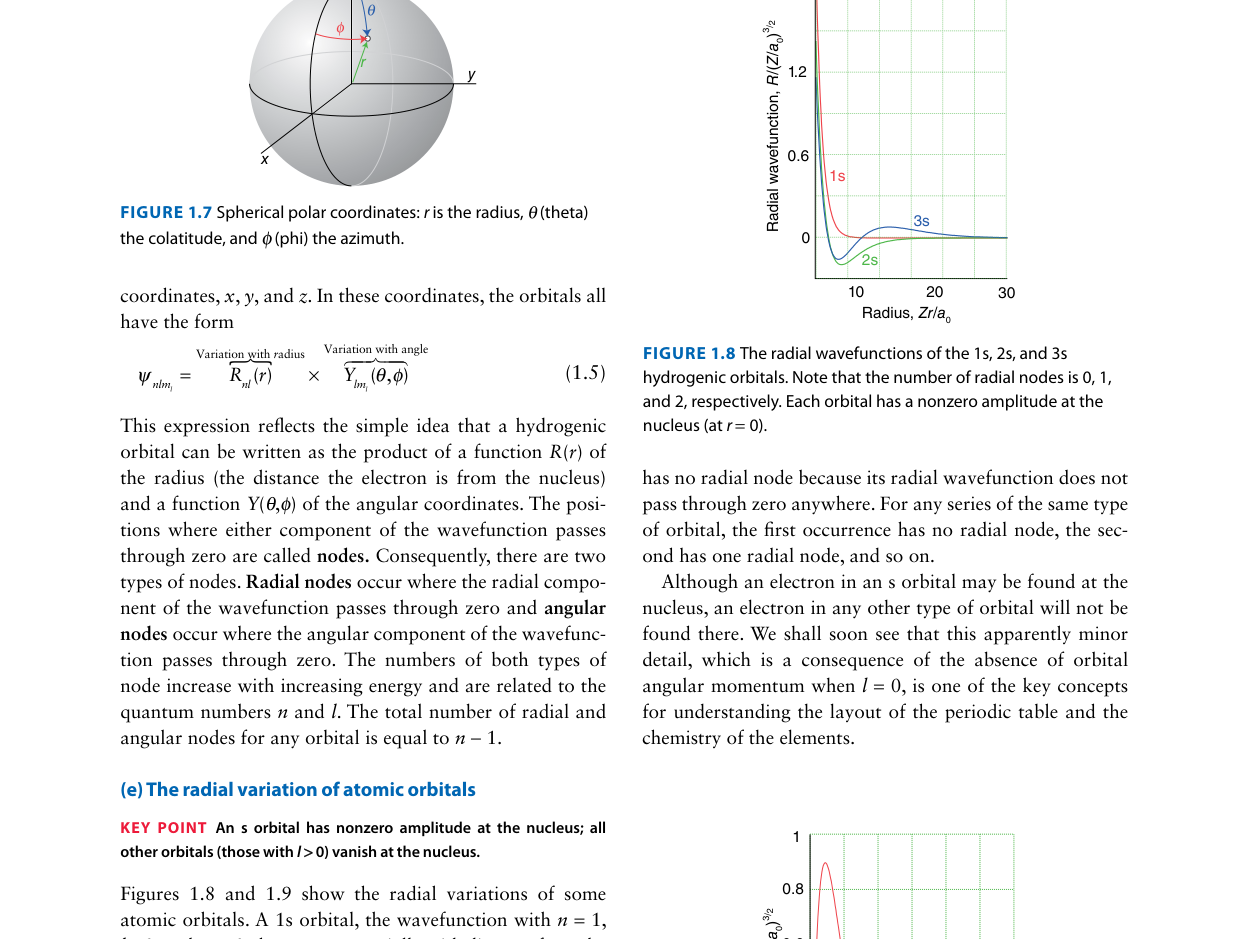
Examine the radial wavefunctions above. The 3s orbital crosses zero (R(r) = 0) at specific values of r. How many radial nodes does the 3s orbital have?
The number of radial nodes is given by (n − l − 1). For the 3s orbital: n = 3, l = 0, so radial nodes = 3 − 0 − 1 = 2. You can see this directly in the figure — R(r) crosses zero twice before approaching zero at large r.
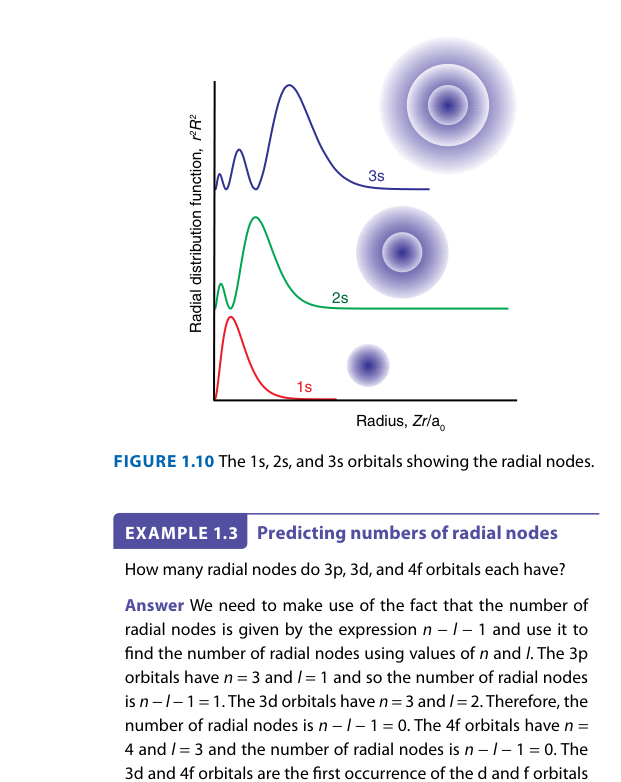
The radial distribution function 4πr²R(r)² differs from the probability density ψ². What does the radial distribution function represent?
While ψ² gives the probability density at a point, the radial distribution function 4πr²R(r)² gives the probability of finding the electron anywhere in a thin spherical shell at distance r. The peak of this function gives the most probable distance — for 1s, this equals the Bohr radius a0.
Quantum Numbers & Orbital Classification
Questions 4–8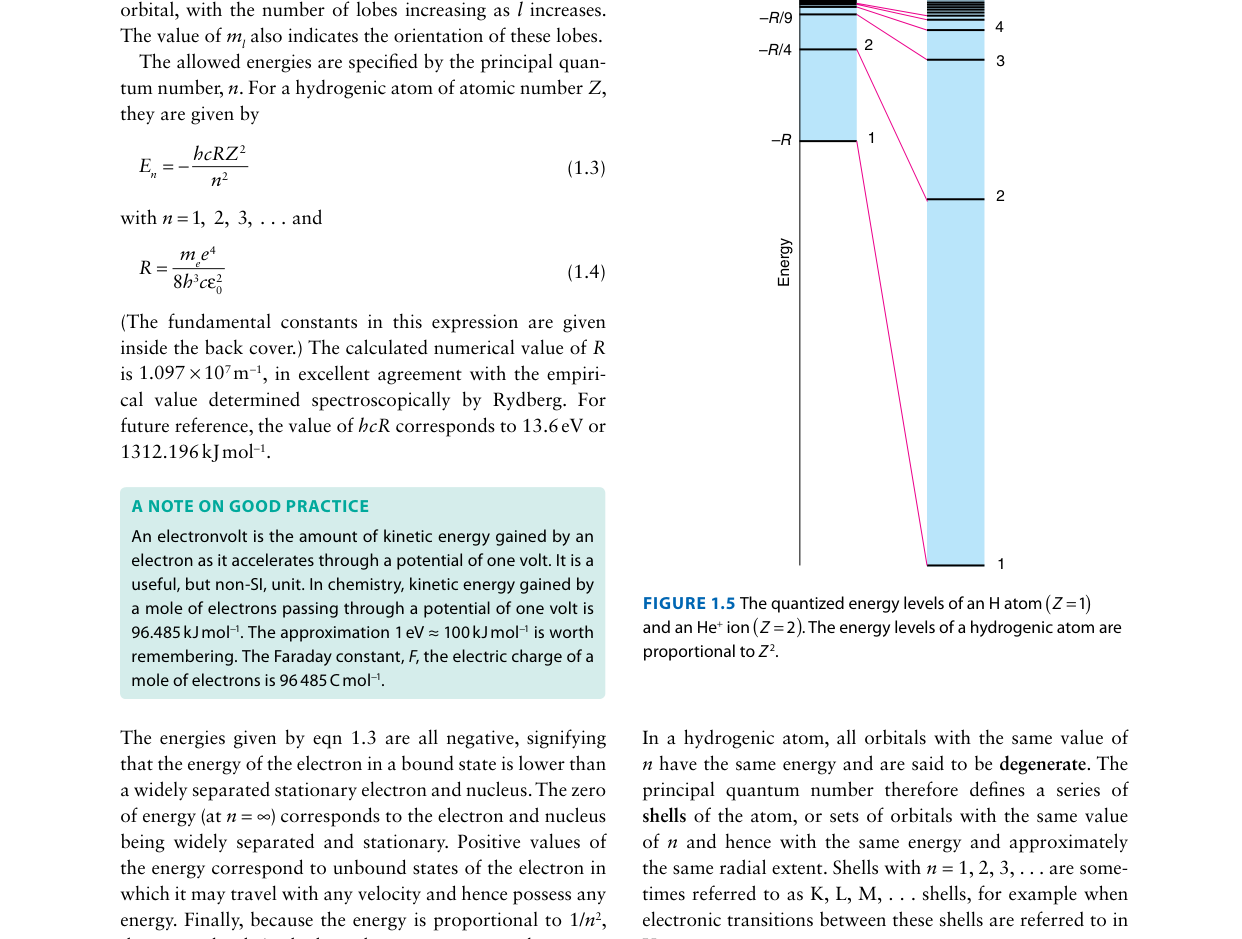
Looking at the energy level diagram for hydrogen above, all orbitals with the same value of n have the same energy. This degeneracy is a special property of which system?
In hydrogen (and any one-electron system), energy depends only on n, so 2s and 2p are degenerate. In many-electron atoms, electron–electron repulsion breaks this degeneracy: orbitals with lower l penetrate closer to the nucleus and are stabilized, so E(2s) < E(2p).

For a given value of l, how many orbitals belong to that subshell?
The magnetic quantum number ml ranges from −l to +l, giving (2l + 1) values. Each value of ml corresponds to one orbital. So: s (l=0) has 1 orbital, p (l=1) has 3, d (l=2) has 5, f (l=3) has 7.
Which set of quantum numbers is not permitted for an electron in an atom?
The orbital angular momentum quantum number l can only take values 0, 1, ..., (n−1). For n=2, the maximum l is 1. So l=2 is not permitted when n=2. This would require at least n=3.
The total number of nodes (radial + angular) in any orbital is given by:
Total nodes = (n − 1). This splits into (n − l − 1) radial nodes plus l angular nodes. For example, a 3d orbital (n=3, l=2) has 2 − 0 = 0 radial nodes and 2 angular nodes, totaling 2 = (3 − 1).

The p orbitals shown above each possess a nodal plane passing through the nucleus. This nodal plane is an example of which type of node?
The nodal plane of a p orbital is an angular node — it arises from the angular part Y(θ,φ) of the wavefunction. Each p orbital has l = 1 angular node (one plane). Similarly, d orbitals (l = 2) have 2 angular nodal surfaces, which may be planar or conical.
Many-Electron Atoms & Penetration
Questions 9–11
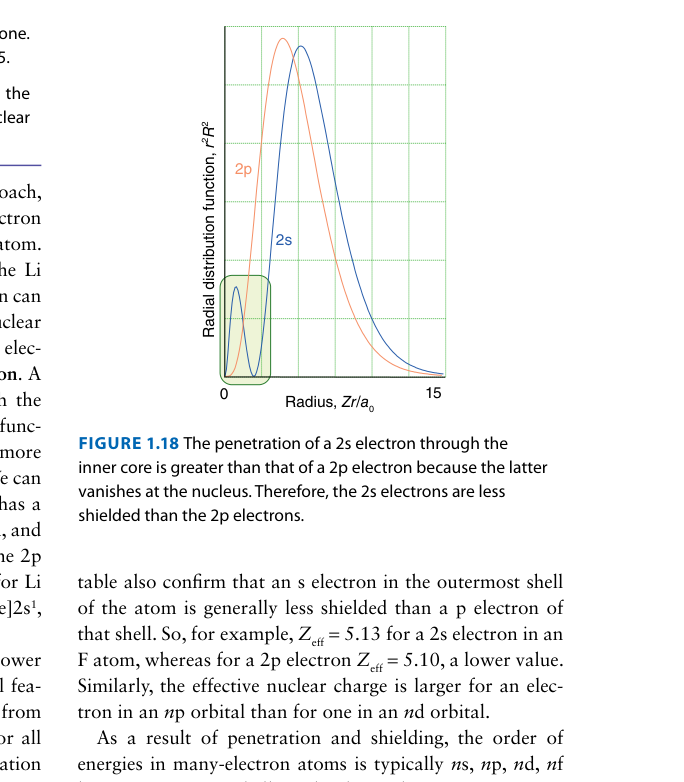
In a many-electron atom, the 2s orbital is lower in energy than the 2p orbital, even though both have n = 2. The figure above illustrates the reason. What is it?
The 2s radial distribution function has a small inner peak close to the nucleus. This means the 2s electron "penetrates" through the 1s shell and experiences a higher effective nuclear charge Zeff than the 2p electron. Greater penetration → less shielding → lower energy. The general rule: for a given n, lower l means greater penetration.
Compare the energy level diagram above for a many-electron atom with the hydrogen atom diagram (Fig. 1.5). What is the key difference?
In hydrogen, energy depends only on n (2s = 2p). In many-electron atoms, electron–electron repulsion lifts this degeneracy: orbitals with lower l penetrate more and are stabilized, so E(ns) < E(np) < E(nd). This is why the aufbau filling order is not simply by n alone.
According to Slater's rules, which of the following correctly describes the shielding experienced by a 3d electron?
Slater's rules assign different shielding contributions depending on the relative positions of electrons. For d and f electrons: same-group electrons shield by 0.35, while all electrons in lower groups (closer to the nucleus) shield by 1.00. This gives the effective nuclear charge Zeff = Z − S.
Periodic Trends
Questions 12–15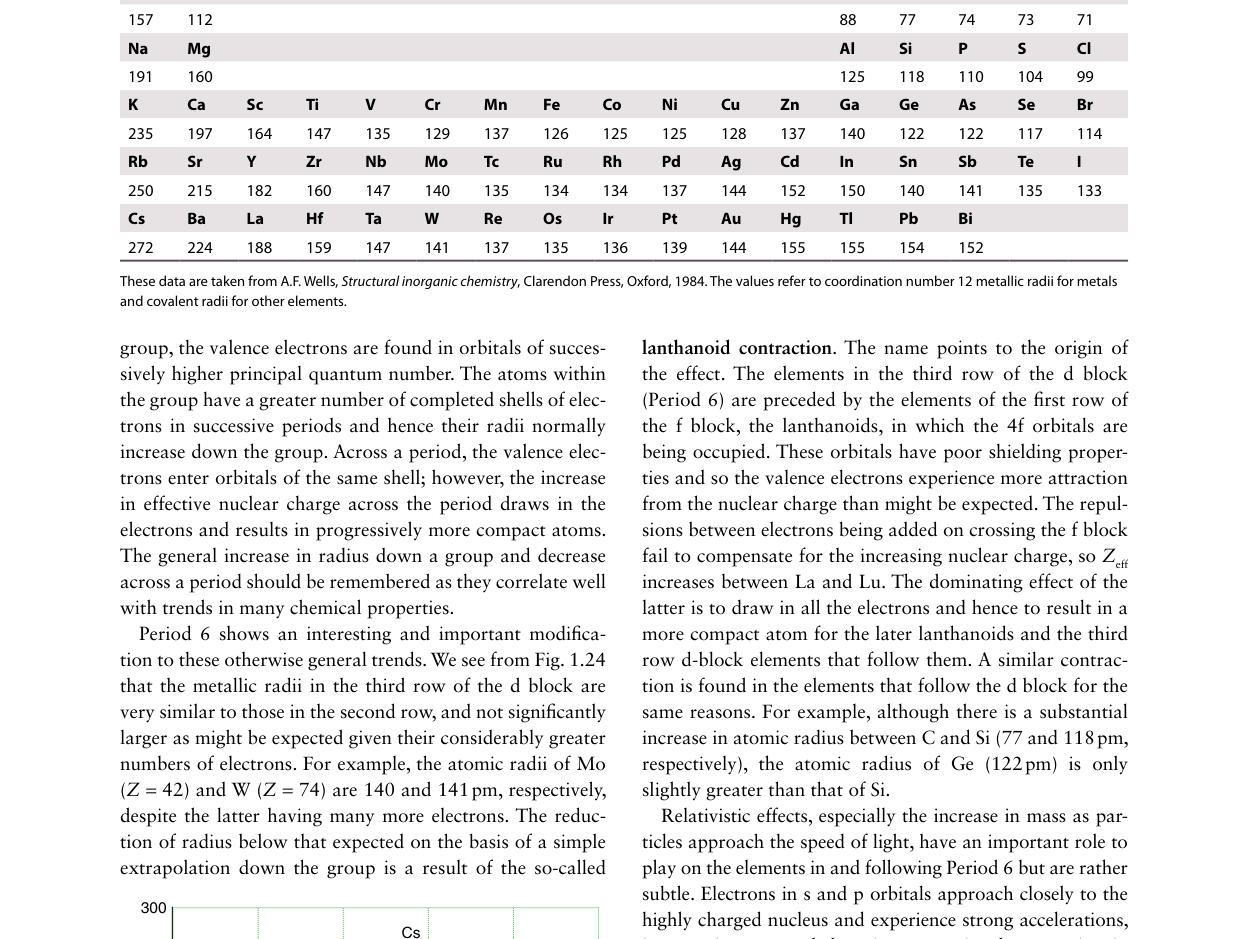
Examine the atomic radius trends in the figure. Across a period (left to right), atomic radii generally decrease. What is the primary reason?
Across a period, protons are added to the nucleus while electrons are added to the same valence shell. Same-shell electrons shield each other poorly (Slater: only 0.35), so Zeff increases steadily, pulling the valence shell inward and decreasing the atomic radius.
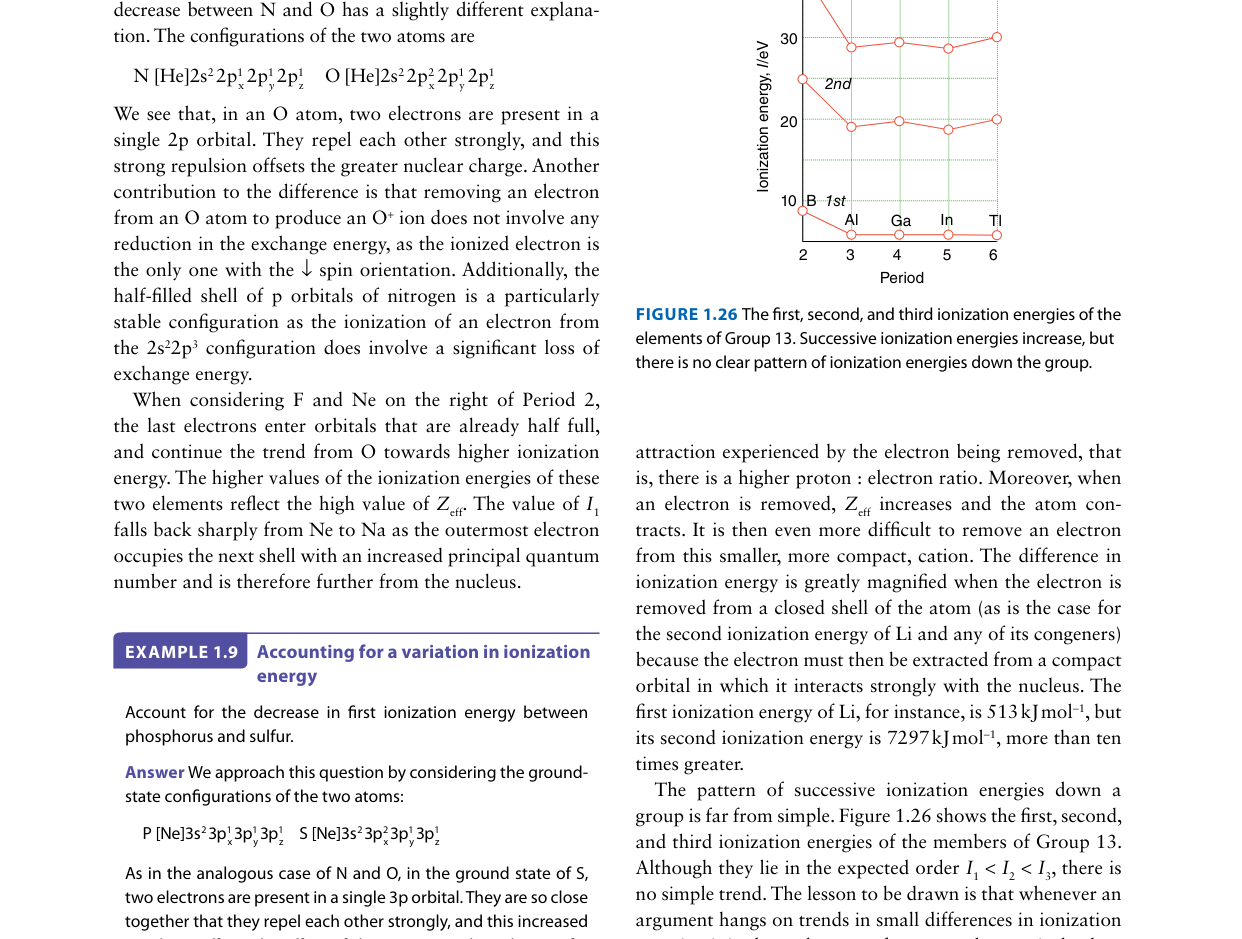
The first ionization energy of oxygen (1314 kJ/mol) is lower than that of nitrogen (1402 kJ/mol), despite oxygen having a higher nuclear charge. Which explanation accounts for this?
Nitrogen's 2p3 configuration has one electron in each 2p orbital (half-filled, all parallel spins). Oxygen's 2p4 forces one 2p orbital to be doubly occupied. The electron–electron repulsion in this paired orbital destabilizes the electron, making it easier to remove — hence the lower IE.
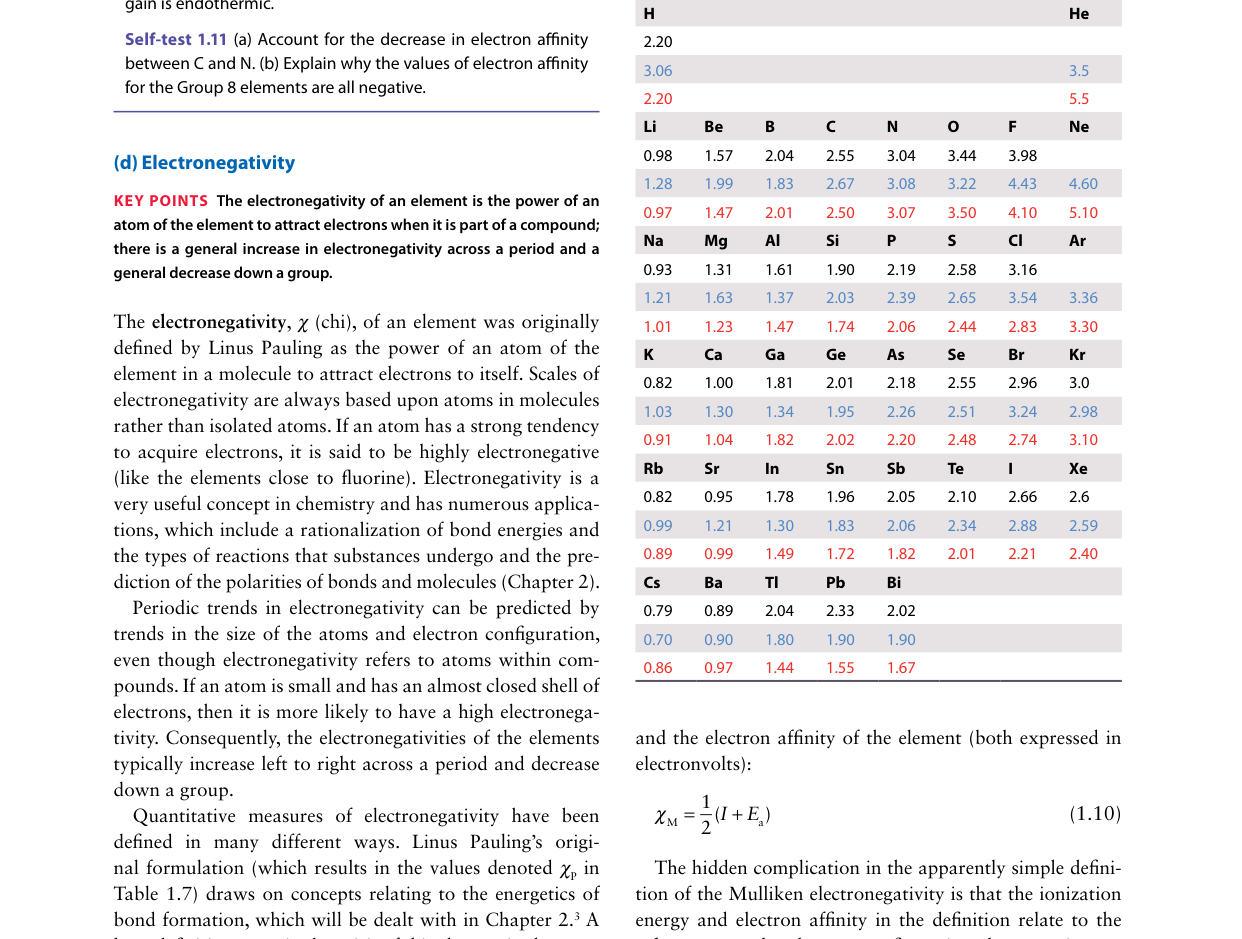
The Pauling electronegativity scale is defined relative to which element?
Pauling defined his electronegativity scale by analyzing bond dissociation energies of heteronuclear diatomics. Fluorine was assigned the highest value (χ = 4.0) as the reference point. All other electronegativities are measured relative to this anchor. The scale ranges from Cs (~0.7) to F (4.0).
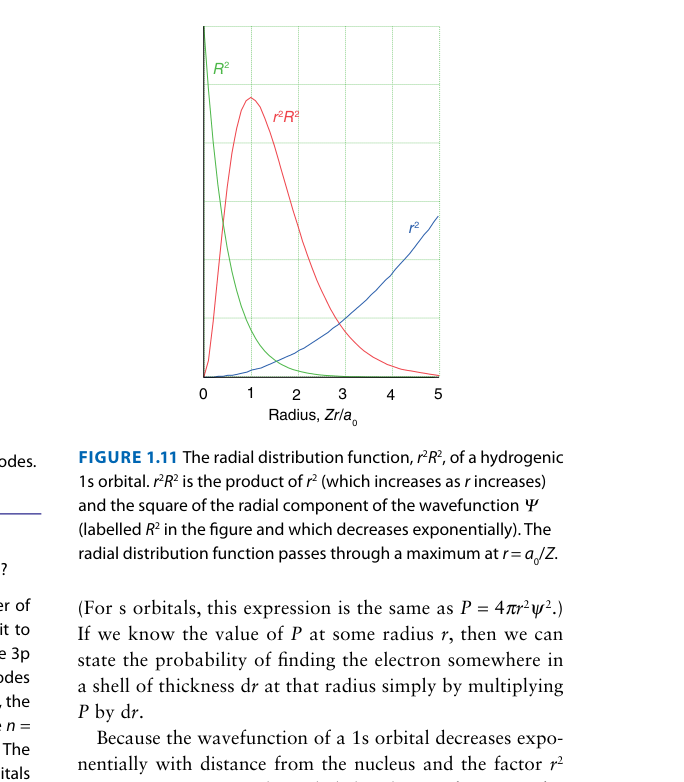
Compare the radial distribution functions of the 2s and 2p orbitals in the figure. Which orbital has greater probability of being found very close to the nucleus?
The 2s radial distribution function shows a small but significant inner peak close to the nucleus. The 2p function has zero probability at r = 0 (it starts from zero). This inner peak is why 2s "penetrates" through inner electron shells and experiences a higher Zeff — directly explaining why E(2s) < E(2p) in many-electron atoms.